 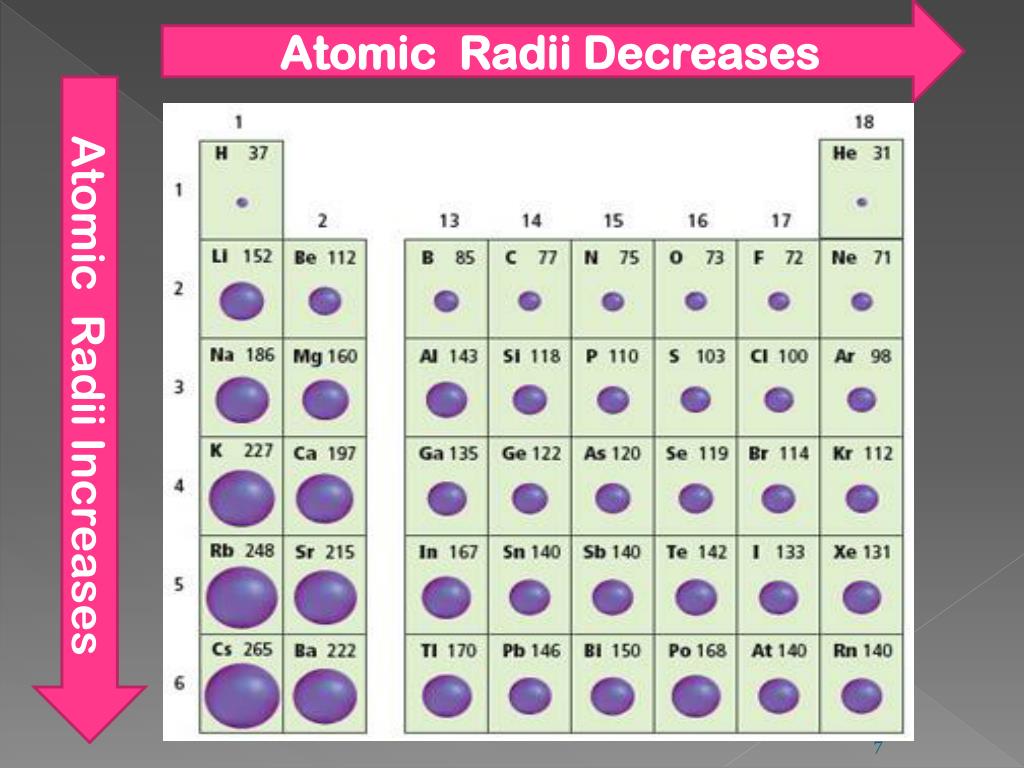 
Image showing periodicity of the chemical elements for atomic radii (Clementi) in a 3D periodic table column style. The Periodic Table helps us remember that the. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a 3D spiral periodic table column style. The atomic radius is half of the diameter of the atom and is a direct result of the number of protons, neutrons, and electrons that compose the atom. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a spiral periodic table heatscape style. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a periodic table heatscape style. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a periodic table cityscape style. Are you a chemistry student Visit A-Level Chemistry to download comprehensive revision materials - for UK or international students Atomic radius. Francium has the largest atomic size on the periodic table, and helium has the smallest atomic size. As shown in the graph below, the atomic radius is largest at the first element in each period, and it decreases down each period. It is also known as the distance measured. There is a correlation between the atomic radii as determined from these calculations and the radii of maximum charge density in the outermost shell of the atom. As electron cloud sizes increase, so do atomic radii. Atomic radius is the distance measured from the centre of the nucleus to the external shell carrying electrons. You should consult reference 1 for full details, but it is not light reading for most people. These values are calculated using self-consistent-field functions (reference 1). This distance between an atoms nucleus and outer electron shell. The values given here for atomic radius are calculated values using methods outlined in reference 1. The atomic radius of a chemical element is a. Sometimes in text books and other sources, the rather vague term "atomic radius" is not defined and in such cases it is therefore not clear what the values actually mean. Atomic radius is the distance from the center of the nucleus to the outermost shell containing electrons. Many references give table of atomic radii. Atomic radius is defined as half the distance between the nuclei in a molecule of two identical elements. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
